The many problems of using IPA for cleaning fiber optics
By Edward J. Forrest Jr., RMS
In the world of fiber optics, there only are two truly critical precision cleaning operations. The first is “prep” before a fusion splice. The other is endface cleaning. Both are essential to deployment of reliable, high-capacity networks. But as new and advanced as modern fiber networks might be, most technicians still are cleaning them with the same fluid used in the 1960s: isopropyl alcohol, also called IPA.
So here’s the headline: To get the most from their investments in advanced fiber equipment, companies must update their cleaning processes and stop using IPA. Over the past 20 years, the precision cleaning industry has developed high-performance products that cost less, are safer to use and perform better than traditional alcohol cleaners. Let’s take a look.
Why clean?
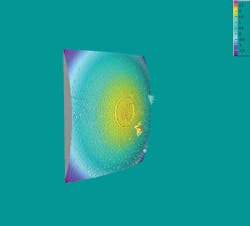
The basic reason to clean is as simple as the stain on your shirt. In fiber optics, this is not merely a cosmetic issue. Stains and debris on an optical surface will degrade the transmission of the signal. “Clean” directly equates to more-reliable transmissions and mechanically stronger splices. Optical fiber must be pristine to transmit reliably and to splice properly.
The existing standard for endface cleaning is confusing. IEC 61300-3-35 clearly states that IPA is no longer acceptable for endface cleaning. But IPA commonly is packaged and promoted for fusion-splice prep. Far too often splicers and endface cleaners forget to send each other “the memo”: Do not use this chemical for every fiber cleaning application.
Throughout the 1990s numerous new solvent choices evolved to replace ozone-depleting chemicals, and some of these experiments migrated into the fiber-optic industry. Some companies used alcohol in different mixtures. Precision hydrocarbons were tried and “tweaked” to minimize environmental issues. New chemicals such as hydrofluoroethers (HFE) and hydrofluorocarbons (HFC) entered the market. I even have seen companies using unconventional cleaning fluids, such as dishwashing detergent and SimpleGreen. It was a crazy time.
Most of the exotic (and ineffective) practices died out due to economic, environmental, performance or worker-safety concerns. Today the list has been abbreviated to just a few, proven choices. Amazingly, more as a nod to tradition than effectiveness, IPA remains on the list. There is another reason: IPA is inexpensive and often purchased from a drug store.
I recall training a fiber-optic distributor and gently suggesting to the owner that IPA was not the best choice for cleaning fiber. Coolly, he advised me that he and his mom had started their massive international business transferring IPA from 55-gallon drums into small bottles. Their highly successful enterprise exists to this day and, “We’re not going to change,” he informed me.
But one has to ask, why not? Why have perceptions of IPA remained unchanged while everything else in the industry has evolved at a furious rate?
What don’t we understand?
Isopropyl alcohol has many names. On safety data sheets it may be called isopropanol, rubbing alcohol, propan-2-ol, 2-propanol, dimethyl carbinol or just IPA. It is an organic compound with the chemical formula C3H7OH. No matter what the name, the safety data sheet will carry the official “CAS number” of 67-63-0. It is a colorless, flammable compound with a strong odor. It has a wide variety of medical, industrial and household uses but it has many flaws as well.
For a start, IPA is neither a very effective nor very versatile cleaner. IPA’s cleaning ability is limited to human body oil and iconic salts but not much more. IPA is not effective on mineral oils and most hand lotions. IPA does not clean oils and greases very well. Because fiber optics have now expanded into mining, oil fields, as well as telecom networks and data centers, selecting “the right stuff” becomes an essential best practice.
Drying speed is another issue with IPA. Slow-drying IPA can be trapped inside a fiber-optic connector. This once-pure liquid may leach out inconveniently and contaminate an endface. A familiar characteristic of any liquid is its boiling point; the lower the boiling point, the faster a liquid will evaporate. Water boils at 100 degrees Celsius and dries slowly; alcohol boils at 78 degrees Celsius so it dries slowly as well. Many companies have introduced new cleaning choices that boil near room temperature. These cleaners will “flash dry” and will not leave contamination or moisture trapped on the fiber.
IPA has another unexpected characteristic; it is hygroscopic, which means it attracts moisture to itself. Studies of 99.9-percent IPA reveal that a bottle of IPA in an open container will lose 7 percent of its strength in as little as 15 minutes. IPA continues to absorb moisture until it reaches equilibrium at about 65 percent. This is why the “rubbing alcohol” purchased at a local store is about 30-percent water. The manner in which the cleaner is packaged is critical to its performance.
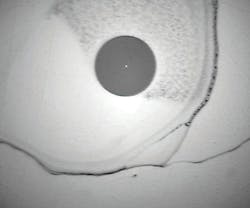
Water doesn’t just dilute IPA’s already-feeble cleaning power. It also will add residues carried in the air. These contaminants from the air-and others from those cheap plastic bottles-will be left behind when the IPA dries. This is the source of the “haze” that IPA leaves after cleaning. The haze may interfere with transmission of light (increased insertion loss) and also may affect the transmission of one wavelength versus another wavelength, which can be very troubling in wavelength-division-multiplexing systems.
No matter how pure the IPA may be when purchased, it will become contaminated when it is poured into a pump bottle, dispenser or an uncovered container. Guaranteed. Each time a drop of IPA is squeezed out of a traditional pump-bottle, air enters. This is reality-not just sometime, not just maybe-but each time it is dispensed. IPA out, water and contamination in. For optimal results, the packaging should be hermetically sealed.
There is another concern: the “headroom” in the drum or pail of IPA will have air that contains moisture. This exposure continues hygroscopic deterioration. There is no practical way a technician can estimate the purity of IPA that has been opened or improperly stored. The best practice is not to use the IPA at all.
Can companies buy pure IPA and solve the haze problem? Many try, but I believe that the purchase of 99.9-percent “reagent grade IPA” is a waste of money and effort. Here’s the little secret about reagent grade IPA: Because it is the most pure it is the most hygroscopic; 99.9-percent reagent-grade IPA will reduce itself to drug-store purity long before the container is emptied. Unless the techs are buying reagent-grade IPA in a hermetically sealed container and using it under very controlled conditions (such as protecting it with a nitrogen blanket to control moisture absorption), your company is just wasting money.
Storage and packaging are problems rarely discussed, but are crucial in this environment. IPA is a hazardous liquid because it is flammable. All flammables should receive special storage and handling. These characteristics also make it difficult and expensive to ship IPA, and IPA certainly cannot be carried onto airplanes because of numerous TSA, DOT, FAA and IATA (International Air Transport Association) regulations. As a result, it often is difficult for techs in remote locations to have the proper cleaning fluid on hand. A better choice would be to select a nonflammable, nonaerosol, nonhazardous liquid, which are easy to ship anywhere.
Environmentally, IPA is a volatile organic compound (VOC). This means it contributes to local smog. Now, tiny bottles of IPA aren’t going to do much damage. But many locales-including California and New Jersey-have implemented clean-air legislation that make it very difficult to legally use VOCs in such containers. Emerging nations such as China have also considered the VOC problem to become a better global neighbor. So while a technician may be able to get IPA it may not be legal to use it. Single-use penalties can be as high as five figures and include incarceration. Yes, we live in a new legislative environment, and one of “best practice” performance.
For fusion splice prep there is one last concern: The residual moisture in the IPA will corrode the electrodes on expensive fusion splicing equipment. As the high-intensity arc fuses the fibers, the heat not only evaporates remaining IPA, but also the moisture embedded in the IPA. Yes, you can replace the electrodes, but would it not be better practice to select a chemical that does not hasten their demise? This is the reason many major fusion-splice producers have gravitated away from IPA for splice prepping and now market application-specific products in unique packages.
What’s the answer?
The point is simple. There have been many important advances in the science of cleaning over the past three decades. The essential reality is IPA has been surpassed and replaced by safer and more-effective chemicals.
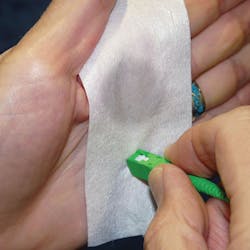
Fiber optics need fast, consistent and reliable contamination removal, as well as a dry surface. The selection of a fluid for cleaning fiber optics is a complex tradeoff of many attributes-cost, storage, handling, purity, actual cleaning ability, drying time and worker safety to name a few. Modern, nonflammable fluids, along with the appropriate non-cellulose wipers (no paper, cosmetic-like nose tissues!), are the best choice to ensure a clean surface that is optimized for ultra-high speed and ultra-capacity for fiber optics. The best choices are ultra-pure, fast-evaporating, nonflammable cleaners in non-refillable, hermetically sealed containers. When choosing a wiper, I prefer hydrogenated non-woven polyester/cellulose cloth, not cheap paper wipes. These are readily available. Just ask.
Look for better products. They are on the same catalog page as IPA. Remember, by the time that expensive reagent-grade IPA is used it will have lost almost all its cleaning ability, wasting time and money, and compromising the performance of your network.
Edward J. Forrest’s career in fiber optics has included the issuance of several patents, development of products in production, and pending patents. He is recognized as an expert on the three-dimensional nature of connectors, contamination, cleaning and the implications for network design and performance. Forrest wrote this article on behalf of Sticklers (www.sticklerscleaners.com). He can be reached via email at [email protected]. The images in this article were supplied by MicroCare’s Sticklers Products.
Fluid for automated endface cleaning
At the European Conference on Optical Communication (ECOC), held in late September in Dusseldorf, Germany, MicroCare Europe featured CleanBlast as part of its Sticklers fiber-cleaning portfolio. Specifically, Sticklers announced that CleanBlast-compatible HFE-based fiber cleaning fluid from Viavi Solutions is now packaged by Sticklers.
“The CleanBlast chemistry is engineered for the automated, high-speed cleaning of fiber endfaces using the CleanBlast systems manufactured by Viavi Solutions,” Sticklers explained. “These systems are widely used around the world by OEMs making transceivers, patch cords and other quality fiber-optic devices. Thousands of systems are in use in Europe, the USA, Mexico and across Asia and every one of these systems needs a supply of the CleanBlast HFE-based cleaning fluid.”
Though not for use in the field by technicians to keep fiber endfaces clean, CleanBlast is an important piece of the fiber-connectivity ecosystem in that it ensures that endfaces are clean when they leave the factory. As of the conclusion of ECOC on September 21, Viavi Solutions no longer offers the cleaning fluid; it now refers customers needing CleanBlast fluids to Sticklers.-Ed.